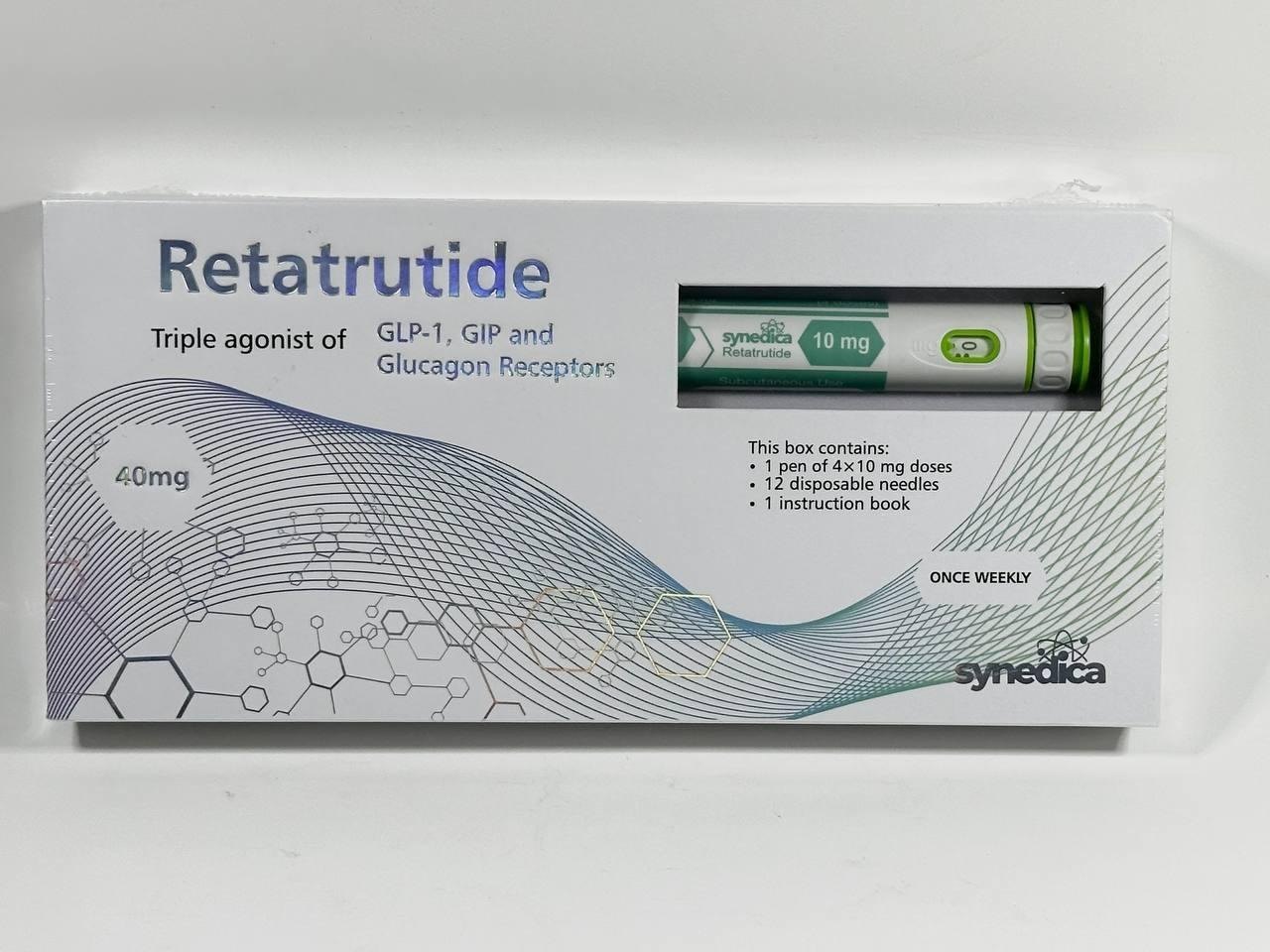
Synedica Retatrutide 40 mg: Definition, Composition and Terminology
What is Retatrutide?
Synedica Retatrutide and Synedica Retatrutide 40 mg are synthetic peptide compounds studied in metabolic and endocrine research. They belong to a class of compounds designed to interact with multiple hormone receptors involved in physiological regulation.
Structurally, retatrutide is classified as a multi-receptor agonist peptide. In laboratory and clinical studies, it is being investigated for its interaction with signaling pathways related to energy balance and metabolic signaling.
Retatrutide is not a dietary supplement, a cosmetic ingredient, or an approved drug. Its use is limited to controlled research and experimental settings.
What does the name “Synedica Retatrutide” mean?
The term Synedica Retatrutide is not a recognized international non-proprietary name (INN) nor is it a registered pharmaceutical brand. It is a combination of a substance name and an identifier used in online environments.
Within peptide and research environments it is common practice that:
-
a supplier name
-
a laboratory identifier
-
or a catalogue reference
is placed before the name of the substance. Over time, this combination becomes a search term, even if it is not an officially registered trademark.
Therefore, Synedica Retatrutide should be understood as a descriptive designation, not as a legal or regulatory classification.
What does “Synedica Retatrutide 40 mg” mean?
The term Synedica Retatrutide 40 mg refers to a quantitative label, not a dosage recommendation.
Important clarification:
40 mg does not mean a single dose, daily dose, or prescribed amount.
In peptide contexts, “40 mg” usually means:
-
the total amount of peptide in a vial, cartridge, or pen
-
a packaging or inventory specification
-
a laboratory or catalog unit
Milligram measurements are often misunderstood by people unfamiliar with research peptides. This text clarifies this intentionally to prevent misinterpretation.
Milligram designation for peptides
For peptides , milligram (mg) values describe mass , not utilization.
For example:
-
“40 mg” means the total amount of peptide
-
It does not indicate concentration after dissolution
-
It does not specify frequency, time or method of administration
This is the standard in research peptides and experimental compounds.
Composition of Retatrutide
Retatrutide consists of a specific amino acid sequence designed to bind multiple hormone receptors. Like other synthetic peptides, it is produced via controlled peptide synthesis to ensure accuracy and purity.
Key features include:
-
a defined amino acid chain
-
molecular modifications for stability
-
high purity requirements
These properties are relevant for laboratory analysis, quality control and research reproducibility.
Retatrutide as a research peptide
Retatrutide is classified as a research peptide . This means:
-
it is used in preclinical and experimental research
-
it is not permitted for sale as a medicine or supplement
-
use is subject to research and laboratory protocols
Any reference to Synedica Retatrutide must be interpreted in this research context.
Commonly used terms
People often encounter these terms:
-
Retatrutide peptide
-
Retatrutide compound
-
Retatrutide research material
-
Retatrutide 40 mg vial or pen
These all refer to the same peptide, just labeled differently.
Common misconceptions
Misconception 1: “40 mg is the dose
.” Incorrect. It refers to the total amount in the package.
Misconception 2: Synedica Retatrutide is an approved drug
. Incorrect. It is not a registered drug.
Misconception 3: Different names mean different substances
. Incorrect. They refer to the same peptide.
Frequently Asked Questions
What is Synedica Retatrutide?
It’s a generic name for retatrutide in certain settings, not an official brand name.
What does 40 mg mean?
It refers to the total amount of peptide in the package, not a dosage.
Is Synedica Retatrutide different from retatrutide?
No. It’s the same substance, just named differently.
Is retatrutide approved for general use?
No. It is an investigational drug and is only permitted in research settings.